
ECA, 90 capsules I VemoHerb®
26,90 €
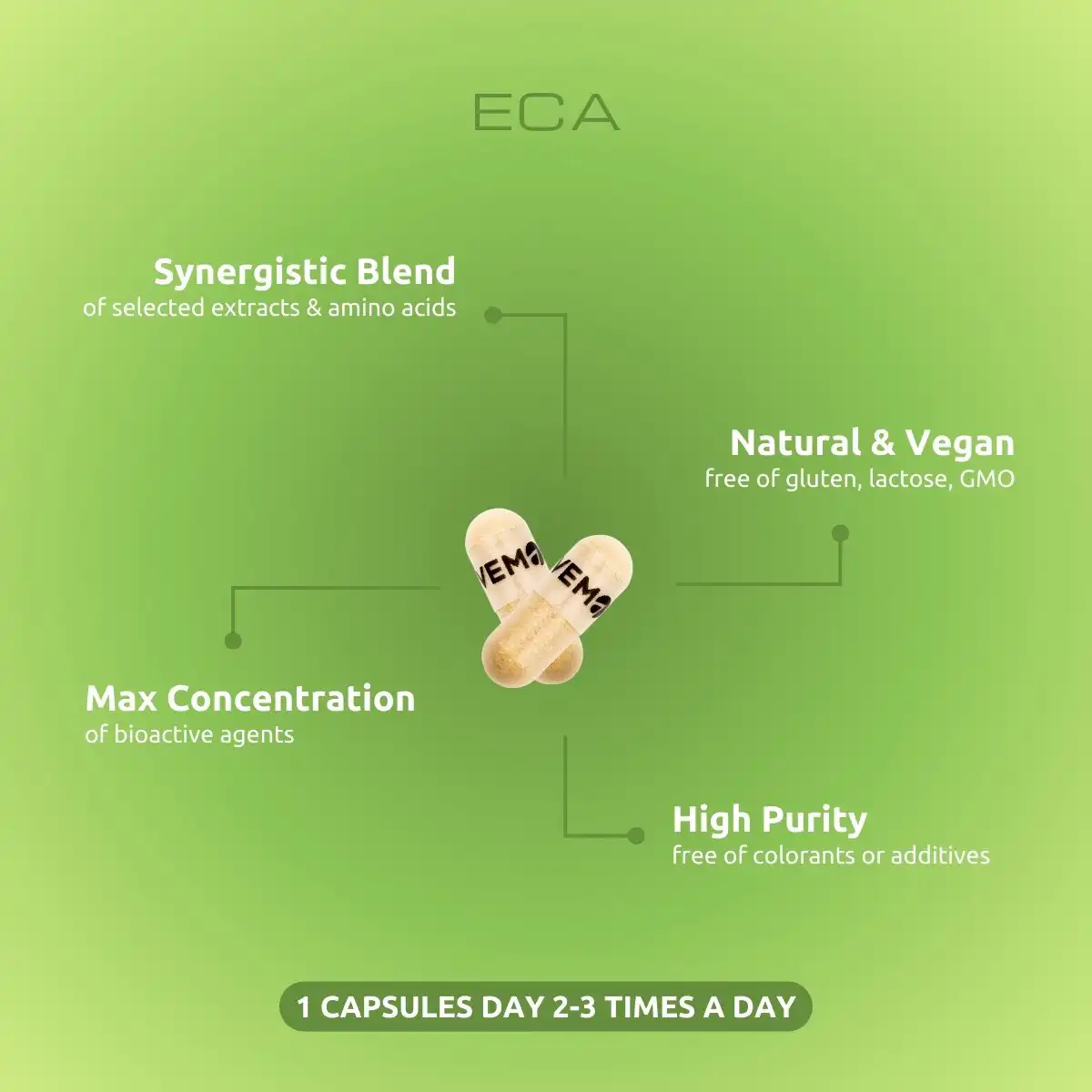
Bioactive blend that helps the body get rid of excess weight, fat and water naturally.
VemoHerb® ECA is a synergistic complex of extracts from White Willow Bark, Bergamot and Caffeine,. This supplement is intended to support fat burning, loss of excess water and increased energy.*
An effective thermogenic stimulant that supports:
- Intensive fat burning*
- Weight and excess water loss during training*
- Endurance and athletic performance during workout*
VemoHerb® supplements are made with standardized extracts for maximum potency and purity. Each product is lab-tested to ensure 100% natural, vegan ingredients with NO additives or banned substances.*
Each pack contains 90 vegan capsules.
The product is a food supplement not a medical drug. The product is not a substitute for a varied diet. Do not exceed the recommended daily dose. It is not recommended for pregnant, nursing women and children!
| *These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease. |
RECOMMENDED DOSE
1 capsule 3 times a day. Take the capsule between the meals or as directed on the label.
More information about the right intake and cycling, you can find HERE.
BIOACTIVE INGREDIENTS
200 mg Caffeine anhydrous;
35 mg dry extract from Citrus aurantium, standardized at min 30% synephrine;
60 mg dry extract from White Willow bark, standardized at min 15% salicin.
HOW AND WHY IT WORKS
VemoHerb® ECA is a complex mixture of Caffeine anhydrous, dry extract from Citrus aurantium, and dry extract from White Willow bark.*
The properties of the active substances in VemoHerb® ECA provide for its beneficial effect.*
Caffeine anhydrous
Caffeine lends a hand to increasing the energy, supports the body’s capabilities for intense training, positively influences the normal nerve impulse transmissions and the weight loss.* [1, 2, 3]
Citrus aurantium (synephrine)
The synephrine in the dry extract of Citrus aurantium is characterized by its ability to interact with adrenoceptors and to maintain the metabolism thus burning excess fats while preserving muscle tissue.* [4, 5] It also positively influences the suppression of appetite in a synergistic manner, along with the other active ingredients, present in VemoHerb® EKA.* [6]
White Willow bark (salicin)
The dry White Willow bark extract present is standardized to salicin, which is a natural source of salicylic acid, enhancing the action of caffeine and synephrine.* [6, 7] Salicin supports the natural regulation of cAMP levels (a natural reaction of the body) and thus the processes of thermogenesis are maintained highly active for a prolonged period of time.* [8]
The blend
Тhe achieved combination of Caffeine anhydrous, dry extract from Citrus aurantium, dry extract from White Willow bark, is proven to be one of the most effective dietary supplements that influences positively the thermogenesis and weight loss processes.* [5, 7, 9]
SCIENCE
References:
1. Erica R Goldstein, Tim Ziegenfuss, Doug Kalman, Richard Kreider, Bill Campbell, Colin Wilborn, Lem Taylor, Darryn Willoughby, Jeff Stout, B Sue Graves1, Robert Wildman, John L Ivy, Marie Spano, Abbie E Smith,Jose Antonio – International society of sports nutrition position stand: caffeine and performance
2. Eric T. Trexler1, Abbie E. Smith-Ryan, Erica J. Roelofs, Katie R. Hirsch, and Meredith G. Mock – Effects of coffee and caffeine anhydrous on strength and sprint performance
3. Adrian B. Hodgson1, Rebecca K. Randell1, Asker E. Jeukendrup – The Metabolic and Performance Effects of Caffeine Compared to Coffee during Endurance Exercise
4. Sidney J. Stohs, Harry G. Preuss, Mohd Shara – A Review of the Human Clinical Studies Involving Citrus aurantium (Bitter Orange) Extract and its Primary Protoalkaloid p-Synephrine
5. Sidney J. Stohs – Safety, Efficacy, and Mechanistic Studies Regarding Citrus aurantium (Bitter Orange) Extract and p-Synephrine
6. Sidney J. Stohs, Harry G. Preuss, and Mohd Shara – A Review of the Receptor-Binding Properties of p-Synephrine as Related to Its Pharmacological Effects
7. Agata Durak and Urszula Gawlik-Dziki – The Study of Interactions between Active Compounds of Coffee and Willow (Salix sp.) Bark Water Extract
8. Khayyal, El-Ghazaly, Abdallah, Okpanyi, Kelber, Weiser D. – Mechanisms involved in the anti-inflammatory effect of a standardized willow bark extract.
9. Nicholas A. Ratamess, Jill A. Bush, Jie Kang, William J. Kraemer, Sidney J. Stohs, Vincenzo G. Nocera, Megan D. Leise, Keith B. Diamond, and Avery D. Faigenbaum – The effects of supplementation with P-Synephrine alone and in combination with caffeine on resistance exercise performance




























Eva –
It’s amazing! You get energy without shaky feeling, and zero appetite. I am thrilled to find this after spending hundreds and hundreds of dollars on junk. Finally, something that works!
J.B –
I love this product! It keeps me energized and focused for work outs and as an added benefit it keeps my hunger at bay! This is a great product!
Fernando Ramirez –
Dear friends at VemoHerb,
Our young athletes (17-20 yo) in Team Ramirez (Norwegian National Team all of them), have used the supplements Able Male and Training BV and Ecdysterone, Rhodiola Rosea and EКA from VemoHerb for 10 weeks, and experience a remarkable performance in training and competition. These micro nutrients have become an important part of our everyday diet. A big thank you for making high quality natural products that really work!
Best regards,
Fernando Ramirez Chief trainer
Robert Jacobsen Nutritionist Team Ramirez
Oslo, Norway