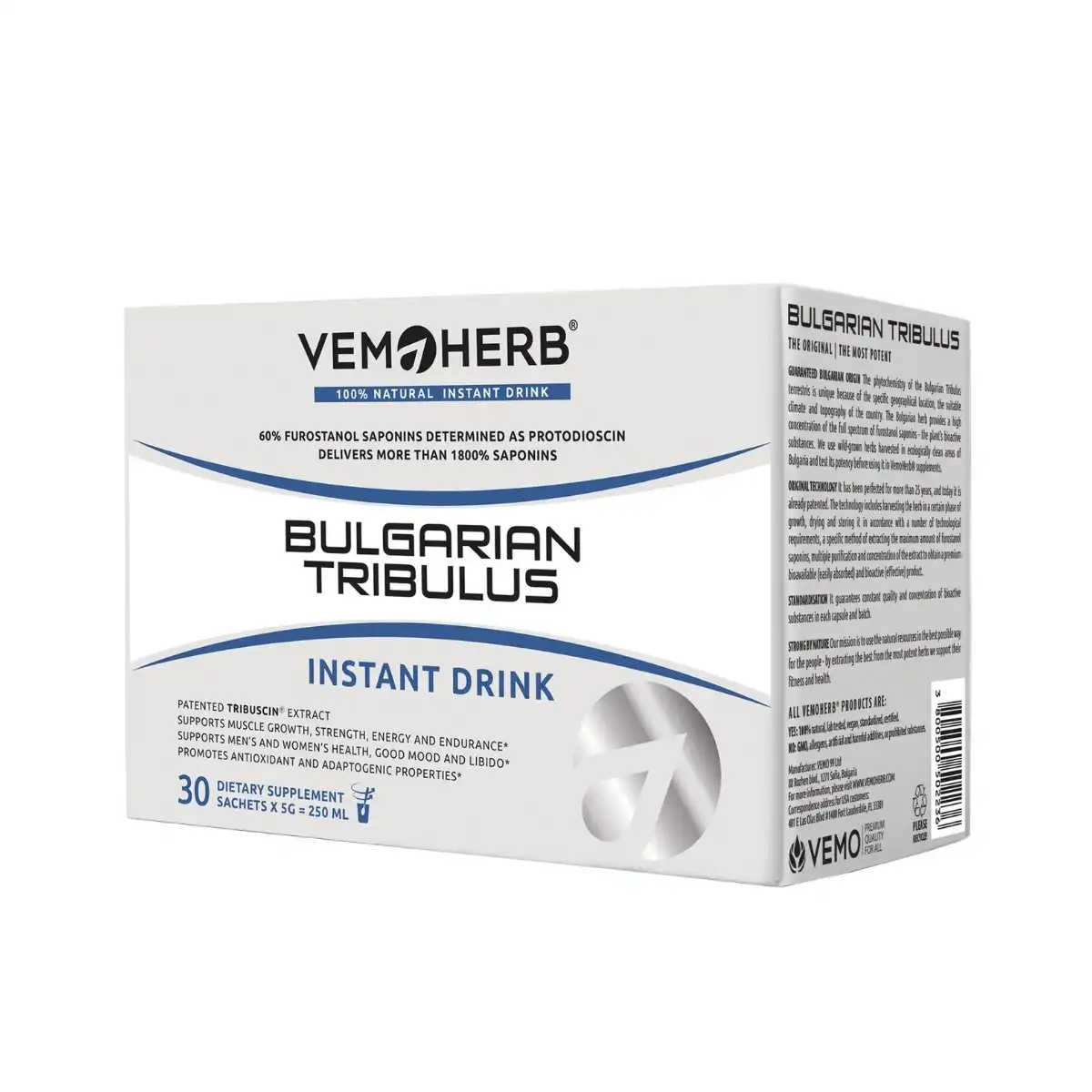
Bulgarian Tribulus Drink, 30 sachets I VemoHerb®
42,90 €
Tribulus Drink with premium quality extract > 1800% general saponins
Completely natural, 100% vegan, instant drink with Bulgarian Tribulus terrestris extract, with flavor.
VemoHerb® Bulgarian Tribulus drink has an energizing effect and assists you to cope with the physical work out.*
Most potent patented Tribulus extract standardized to 60% furostanol saponins (as protodioscin). Delivers more than 1800% general saponins.
Patented as VemoHerb Tribuscin® , this is the first Tribulus terrestris extract in the world completely purified from EU-banned pyrrolizidine alkaloids.
VemoHerb® supplements are made with standardized extracts for maximum potency and purity. Each product is lab-tested to ensure 100% natural, vegan ingredients with NO additives or banned substances.
Each pack contains 30 vegan sachetes.
The product is a food supplement not a medical drug. The product is not a substitute for a varied diet. Do not exceed the recommended daily dose. It is not recommended for pregnant, nursing women and children!
| *These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease. |
RECOMMENDED DOSE
1 sachet 2 times a day. Take between the meals or as directed on the label.
More information about the right intake and cycling, you can find HERE.
BIOACTIVE INGREDIENTS
500 mg Tribulus (aerial parts and fruit) Extract (Tribulus terrestris L.) (60% furostanol saponins determined as protodioscin).
HOW AND WHY IT WORKS
The instant drink VemoHerb® Bulgarian Tribulus contains extract from the Bulgarian herb Tribulus terrestris. This plant is well known in the Bulgarian traditional medicine. Regarded as a vitality herb, it has been used for centuries because of its positive effect on the human energy and libido.* [1, 3, 4, 6, 7, 12]
The Bulgarian Tribulus terrestris extract manufactured by VEMO 99 Ltd is rich in active ingredients and standardized at 60% furostanol saponins determined as Protodioscin. [1, 13] The specific bouquet of furostanol saponins in the Bulgarian Tribulus terrestris have a distinct positive effect on strength, muscle growth, lean body mass, sexuality.* [1, 2, 4, 6, 7, 12]
The main difference between VEMO’s Tribulus extract and all others is not just the higher concentration of protodioscin, but also the higher amounts of accompanying substances that modulate and amplify the effects of Protodioscin. [1, 5]
The whole group of furostanol saponins present in the extract play an important role in supporting the properties of Protodioscin. [1, 5] According to some scientists, the plant extract supports the luteinizing hormone synthesis and positively influences the amounts of testosterone in a natural way.* [8] Testosterone promotes muscle growth indirectly by stimulating protein synthesis in the organism.* [1, 2]
The phytochemistry of the Bulgarian Tribulus terrestris is unique due to the specific geographic position, the suitable climate and topography of the country and the specific collecting method for the herb. These factors determine the differences in the pharmacological and physiological properties and advantages of the Bulgarian herb when compared to Tribulus terrestris with foreign origin. [1, 13]
Most products, available on the market at the moment, contain extracts from India and China, with lower concentration of active ingredients and practically no physiological effect. [13]
The Bulgarian Tribulus terrestris extract assists the positive nitrogen balance and improves protein synthesis, including in the muscles.* This helps with the body capabilities and endurance in cases of physical and mental overwork.* [1, 2]
The extract VemoHerb® Bulgarian Tribulus of VEMO 99 Ltd is different from the rest extracts mainly by:
- Using top-quality wild-grown herb, gathered at a specific phase of growth in ecologically clean regions. The herb is dried and stored according to all technological requirements to guarantee preservation and maximum content of its active substances.
- Unique know-how technology for obtaining potent herbal extract from the plant raw material that provides 60% furostanol saponins with prevailing component Protodioscin.
- Offering a refined herbal product with guaranteed constant quality, containing suitable combination of furostanol saponins for our clients.
SCIENCE
References
1. Wenyi Zhu, Yijie Du, Hong Meng, Yinmao Dong and Li Li – A review of traditional pharmacological uses, phytochemistry, and pharmacological activities of Tribulus terrestris
2. Rogerson, Riches, Jennings, Weatherby, Meir, Marshall-Gradisnik – The effect of five weeks of Tribulus terrestris supplementation on muscle strength and body composition during preseason training in elite rugby league players.
3. K. Milasius, R. Dadeliene, Ju. Skernevicius – The influence of the Tribulus terrestris extract on the parameters of the functional preparadness and athletes’ organism homeostase
4. Jameel Mohd, Ansari Javed Akhtar, Ali Abuzer, Ahamad Javed, Ali M., Tamboli Ennus – Pharmacological scientific evidence for the promise of Tribulus terrestris
5. Mrinalini Kumari, Praveen Kumar and Poonam Singh – Safety Evaluation of Tribulus Terrestris on the Male Reproductive Health of Laboratory Mouse
6. A. Adimoelja – Phytochemicals: modern views and breakthrough in traditional herbal management of sexual dysfunctions
7. A.W. Nasution – Effect of Tribulus terrestris treatment on impotence and libido disorders
8. Milanov, S., E. Maleeva, M. Tashkov. – Tribestan effect on the concentration of some hormones in the serum of healthy subjects (1981) (Company documentation).
9. Justino Mameri FilhoI; Mauro Abi HaidarII; José Maria Soares JúniorIII; Edmund Chada BaracatIV – Effects of the association of estrogen and androgen in postmenopausal women
10. de Souza, K., Vale, F. and Geber, S. – Efficacy of Tribulus terrestris for the treatment of hypoactive sexual desire disorder in postmenopausal women: a randomized, double-blinded, placebo-controlled trial. Menopause (2016) 23: 1252.
11. Postigo, Lima, Yamada, dos Reis, da Silva, Aoki. – Assessment of the Effects of Tribulus Terrestris on Sexual Function of Menopausal Women.
12. Saurabh Chhatre, Tanuja Nesari, Gauresh Somani, Divya Kanchan, and Sadhana Sathaye – Phytopharmacological overview of Tribulus terrestris
13. Dragomir Dinchev a, Bogdan Janda b, Liuba Evstatieva c, Wieslaw Oleszek b, Mohammad R. Aslani d, Ivanka Kostova – Distribution of steroidal saponins in Tribulus terrestris from different geographical regions























Martin Becker –
This stuff is EXCELLENT….. I have been using Bulgarian Tribulus for at least 15 years… This is the most pure and potent Tribulus that I have found. I use it in conjunction with DHEA and Yohimbe and I have Great Workouts, but also on non workout days it gives me increased Stamina and I DO NOT GET as Tired after A LONG DAY.
Jerremy –
I am pleased that this is all natural and not filled with synthetic fillers. Moreover, you’re letting your body to increase your testosterone instead of having a foreign substance introduce testosterone, it is essentially encouraging and sending the signaling for your body to do the work. I have seen definite aesthetic improvements in my own muscle development, and have more definition in my muscles since taking it.